Spravato (esketamine)
The FDA has now approved the first drug that can relieve depression in hours instead of weeks. In a recent article titled “FDA Approves esketamine Nasal Spray For Hard-To-Treat Depression,“ NPR stated:
A study published in the Journal of Clinical Psychiatry found that treatment with Spravato plus an oral antidepressant was associated with a rapid improvement in symptoms of depression, hopelessness, and suicidality within 24 hours of administration.

Spravato
Spravato is a new FDA-approved medication that works for major depressive disorder and treatment-resistant depression. Studies show that Spravato works to reverse depressive symptoms, despite the cause. Studies also show that Spravato is working in every patient category, meaning all ages, genders, races, and duration of depression. Whether it is depression from trauma, menopause, post-partum, life events, or any other reason, Spravato is showing incredibly high rates of remission across the board for everyone. Spravato is working when nothing else has, and in ways other antidepressants never could.
Major depressive disorder and treatment-resistant depression is reversing into complete remission with Spravato nasal spray. Despite who you are or how you got to where you are, Spravato offers hope in better days.
Spravato Dosing for Treatment-Resistant Depression

“This is potentially a game-changer for millions of people,’ said Dr. Dennis Charney, Dean of the Icahn School of Medicine at Mount Sinai in New York. ‘It offers a lot of hope.”
Recommended Treatment Protocols and Pricing
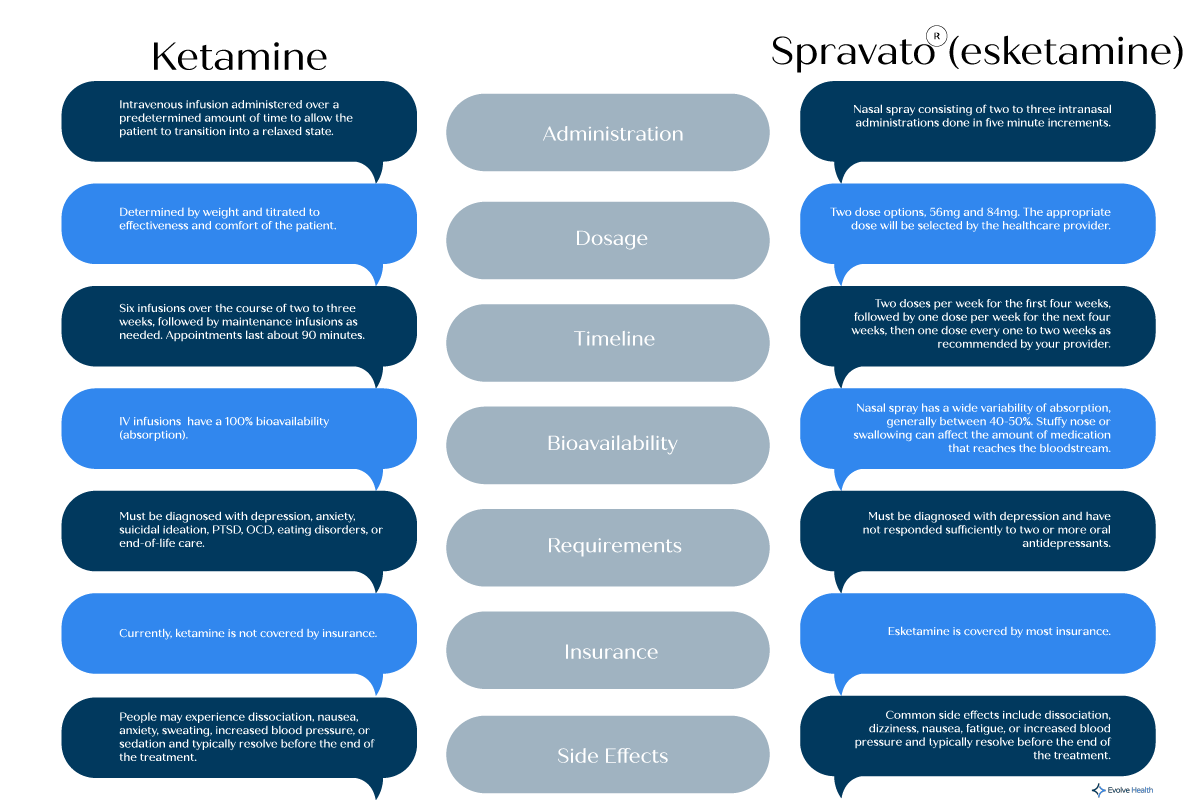
Most insurance companies are now covering Spravato therapy; with a referral, our insurance coordinator can assist with your preauthorization. FDA guidelines state twice a week for four weeks and then weekly for weeks 5-8 and then move to once every one week or two-week dosing. The Spravato website has a nice graphic idea that might help patients who are visual learners.

Referral Forms
FAQs
How to get started
1) Get Pre-Qualified
2) Get a referral
a) From us (on our website) and give it to your doctor.
b) From your doctor and give it to us.
3) Schedule your consultation
What is a referral?
A referral is simply the paper form of your doctor’s approval for treatment with us. You can download a referral above. Your insurance will require a referral, so it’s best to just start there. You can always download a referral form by clicking above. Give it to your doctor and have the discussion about Spravato for help with your depression.
OK, my provider sent a referral. Now what?
Patience and follow up. We will work with your insurance company to get your treatment approved. This process may take a few weeks, so please be patient in the process. Follow up with questions. A few weeks will go by quickly and staying in communication with us is important to starting treatment on time.
I got a denial letter from my insurance company. Will I be able to get treatment?
If all goes well, yes, you can still get treatment. We appeal all denials, and our team will work with your insurance company to get the denial reversed.
How long until it works?
- Variable from patient to patient.
- Patients typically improve within the first 8 weeks; some even have remission of depressive symptoms in this time period.
- Recommend > 6 months trial before determining efficacy outcome.
- Expectation management: Length of treatment varies but is 12-18 months on average.
- Rule of thumb – The longer the depressive symptoms have been present may relate to the length of treatment required.
Patient Should Know
Must have transportation to and from treatment.
Continue taking an oral antidepressant.
Dissociation is not necessary for medication to provide efficacy.
Establishing with a therapist potentially improves outcomes. Treatment works much better for those who also participate in therapy.
The authorization process can take 4-8 weeks. The patient may get a letter of denial initially, but we appeal all denials and typically get them overturned.



